Researchers synthesize new compound, RPI-GLYT-82
May 5, 2026
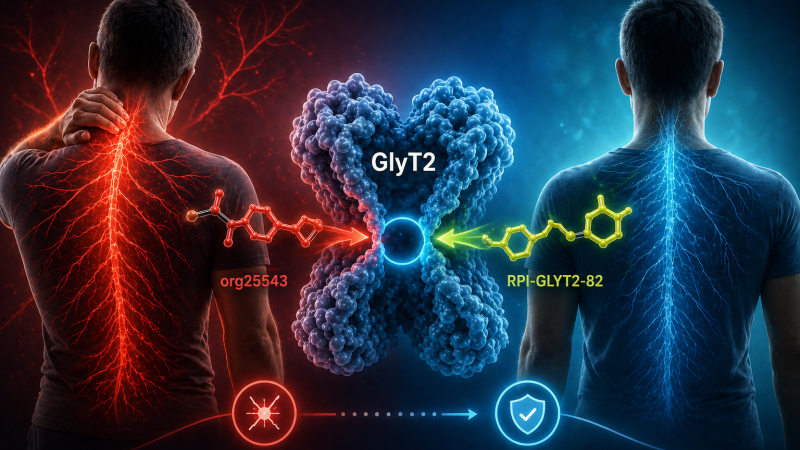
Chronic neuropathic pain remains one of the most challenging conditions to treat, with current therapies offering limited benefit and being over-reliant on opioids. Researchers at Rensselaer Polytechnic Institute (RPI), along with collaborators from the University of Sydney and the University of Copenhagen, have developed an approach that offers a clearer path towards safer, non-addictive pain therapies.
The research, published in Nature Communications, introduces a fundamentally different approach to pain treatment: instead of directly blocking pain signals or relying on opioid pathways, it enhances the nervous system’s natural inhibitory controls.
The approach targets GlyT2, a neuronal glycine transporter. While GlyT2 offers a promising new target for analgesics, most inhibitors cause significant side effects, in part due to irreversible inhibition at analgesic doses. To solve this problem, researchers at RPI have synthesized a new compound designed to bind GlyT2 reversibly — RPI-GLYT2-82 — and they identified its binding site by determining cryo-electron microscopy (cryo-EM) structures of GlyT2.
In preclinical studies, RPI-GLYT2-82 produced effective pain relief without neuromotor impairment, seizures, or signs of addiction — even at doses five times higher than those required for analgesic effect. Cryo-EM structures also captured the transporter in four key conformational states, including when bound to RPI-GLYT2-82 and to the earlier inhibitor ORG25543. These structures revealed an allosteric binding pocket and explained how tightly binding inhibitors can effectively shut down GlyT2 function, leading to side effects.
“We discovered that subtle chemical differences can cause inhibitors to grip the pocket far more tightly and for far longer, effectively eliminating GlyT2 function rather than moderating it,” said Christopher L. Cioffi, Ph.D., co-author on the paper and associate professor in the Department of Chemistry and Chemical Biology at RPI. "This work shows not only that GlyT2 can be safely targeted, but gives us the structural roadmap to do it better."
That roadmap is already being applied. In a follow-up study published in the Journal of Medicinal Chemistry, the RPI team developed a series of next-generation GlyT2 inhibitors based on the same reversible, noncompetitive mechanism. These compounds demonstrated varied potency and recovery kinetics, enabling researchers to further tune how strongly and for how long the inhibitors act. The findings establish a foundation for continued optimization of GlyT2-targeting compounds toward viable preclinical drug candidates.
“These results provide a framework for designing the next generation of compounds with improved pharmacological properties,” said Tanmay K. Pati, Ph.D., research scientist at RPI and co-author on the study. “Our goal is to translate this level of structural and mechanistic understanding into therapies that are both effective and practical for real-world use.”
Working in collaboration with Pati and Cioffi on both papers were University of Sydney’s Robert Vandenberg, Ph.D., Sarasa Mohammadi, Ph.D., and Irina Lotsaris, along with University of Copenhagen’s Azadeh Shahsavar, Ph.D., and Ryan Chantwell Carter.